Prevent
Water generation process & microbial contamination
The prevention of a microbial contamination is crucial within the water generation for the pharmaceutical industry. But how can we persistently prevent such? By understanding the process and thus being able to estimate the contribution of the risk for a microbial contamination for each individual process step. The design of the plant and the maintenance procedures are the two crucial aspects. We need to understand where the germs come from, therefore it is crucial to know how the entire water generation process is structured.
As the EU GMP Annex I states: "... The sterility test applied to the finished product should only be regarded as the last in a series of critical control measures by which sterility is assured. It cannot be used to assure sterility of a product that does not meet its design, procedural or validation parameters. The test should be validated for the product concerned..."
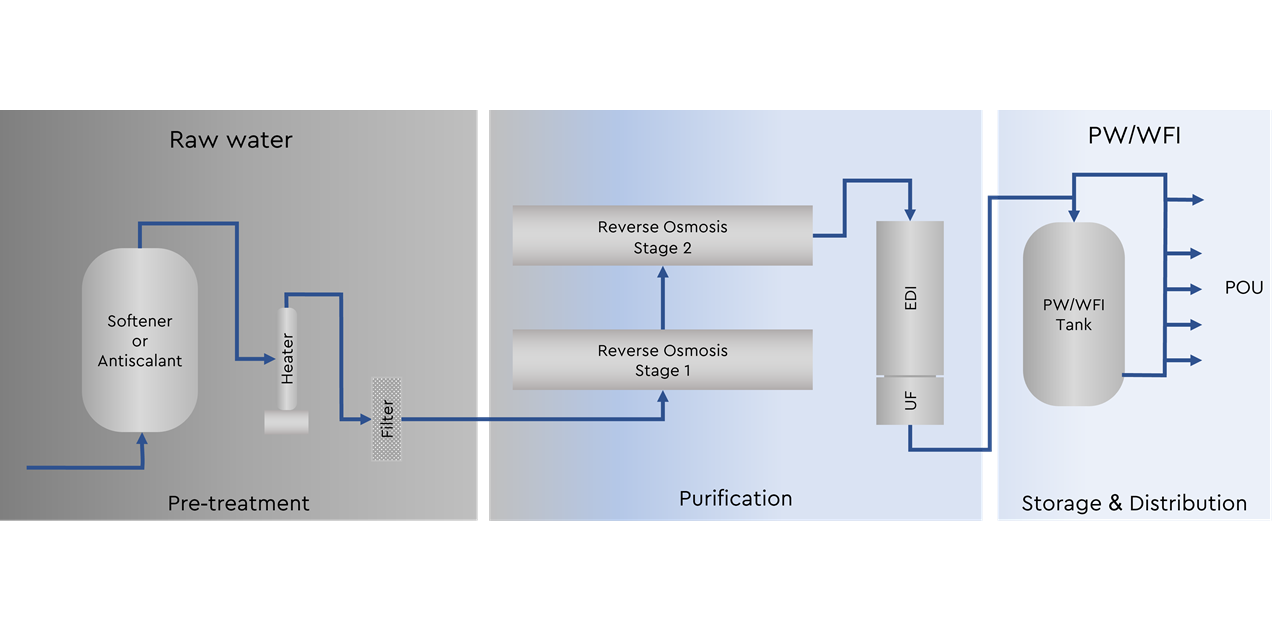
The water generation process in 3 steps
For every step, the contribution to the risk of microbial contamination must be identified. This also applies to maintenance and repair activities. BWT has designed their instruments considering all aspects like minimized dead volumes, sanitizable components, and e.g. special regeneration procedures for the softener to avoid dead zones.
Where do germs come from?
POINT OF USE (POU)
Tank
ELECTRODEIONIZATION (EDI) / ULTRAFILTRATION (UF)
The seal-free spiral design of the septron is the ideal protection against microbial contamination.
Filter
Reverse Osmosis permeate side
Reverse Osmosis concentrate side
Softener
RAW WATER
To guarantee highest microbial safety, regular preventive or better predictive maintenance is recommended. BWT helps you with this.
Pre-Treatment
Various water parameters influence the effectiveness and efficiency of a PW and WFI generation system. Factors such as particles, hardness, chlorination, and other characteristics of the raw water need to be considered when designing a pharmaceutical water generator.
There are several pre-treatment procedures available, such as Softener, Filtering, Antiscalant, and Bisulfite. The softener is broadly used in the pre-treatment process to reduce the concentration of hardness minerals (such as calcium and magnesium) to guarantee that the downstream water treatment processes, in particular the reverse osmosis can operate efficiently. As an alternative antiscalant dosing may replace the softener by increasing the solubility of critical minerals.
Water Purification
The water purification process in cold systems is based on 3 main steps:
storage and distribution
PW or WFI must be distributed from the water generator to the point of use. For storage and distribution, there are several strategies to ensure and maintain the quality.
- Hot storage
- Continuous Electrolytic Ozonization
Want to know more?
Learn more about Contamination Control Strategy and the 4 elements of it...
Unexpected error. Please try to reload the page














